FDA approves Eli Lilly’s Foundayo™ (orforglipron), the first daily GLP-1 weight loss pill with ZERO food or water restrictions. Read the 2026 expert guide.
For the last three years, the weight loss industry has been dominated by a single, unavoidable truth: if you wanted serious, medical-grade results, you had to embrace the needle. Today, that era officially ends. With the FDA approval of Eli Lilly’s Foundayo™ (orforglipron), the rules of metabolic health have been completely rewritten.
Why is this dominating headlines in 2026? Because Foundayo isn’t just another GLP-1 pill. It is the first and only once-daily oral weight loss medication that demands absolutely zero food, water, or fasting restrictions. You wake up, take the pill whenever it fits your schedule, and get on with your life.
Whether you’re a patient tired of weekly injections, a healthcare professional evaluating the next line of obesity care, or an investor tracking Eli Lilly’s market dominance, this comprehensive guide covers everything you need to know about the Foundayo approval.
💡 What is Foundayo™ (Orforglipron)?
Foundayo (generic name: orforglipron) is a once-daily, oral non-peptide glucagon-like peptide-1 (GLP-1) receptor agonist developed by Eli Lilly. Approved by the FDA for chronic weight management in adults with obesity or overweight with at least one weight-related comorbidity, it works by mimicking the GLP-1 hormone to signal fullness to the brain, slow gastric emptying, and regulate appetite.
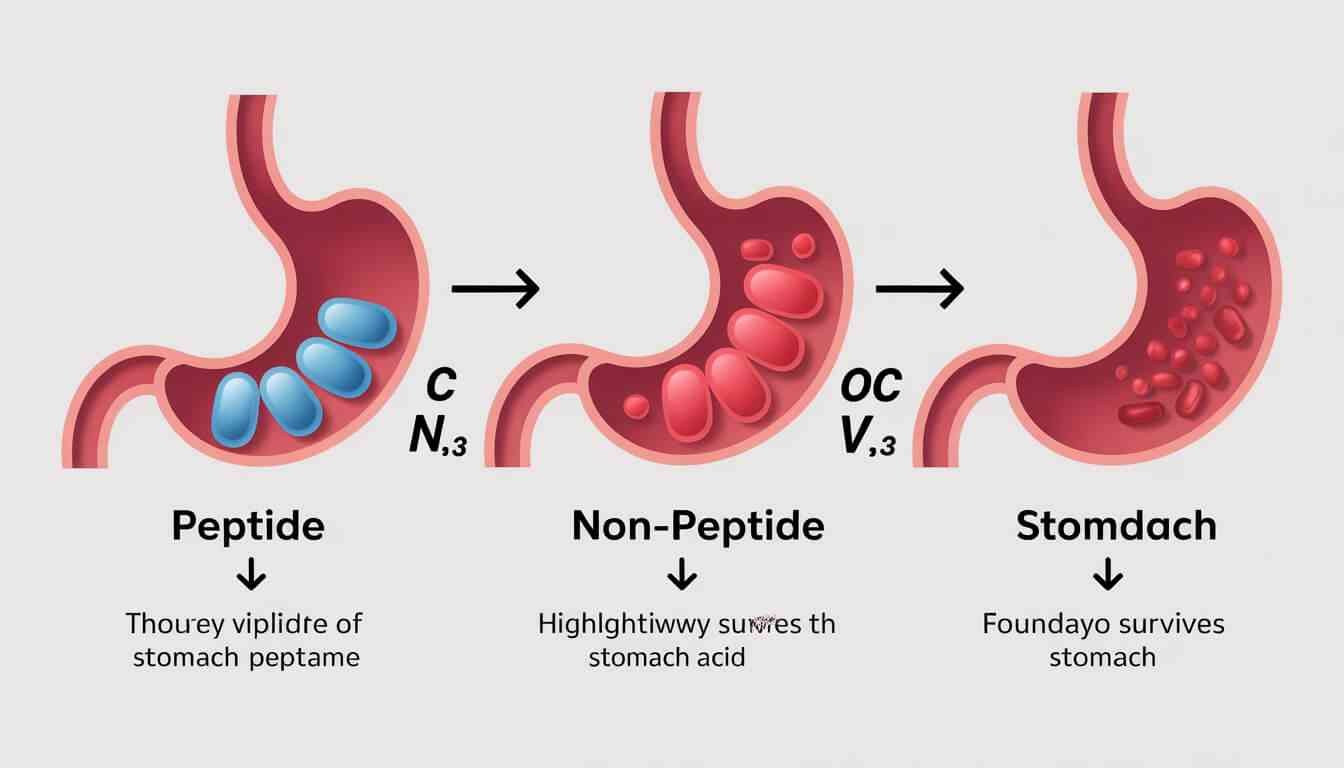
Unlike previous oral GLP-1s (like Rybelsus), Foundayo’s unique synthetic structure allows it to be absorbed in the stomach regardless of food or liquid intake.
The primary keyword here is convenience. Patient compliance in weight loss treatments drops by roughly 30% when fasting rules or injections are involved. Foundayo eliminates this friction entirely.
🔬 The Science: Why the “No Fasting” Rule is a Game-Changer
To understand the magnitude of the Foundayo FDA approval, we have to look at the previous gold standard for oral GLP-1s: semaglutide (Rybelsus).
Rybelsus is a “peptide.” Peptides are fragile. If you take them with a heavy breakfast or a large glass of water, your stomach acid destroys the medication before it can be absorbed. That’s why patients are strictly instructed to take it on an empty stomach with exactly 4 ounces of water and wait 30 minutes before eating or taking other meds.
Foundayo is a non-peptide small molecule. It is synthetically designed to survive the harsh, acidic environment of the human stomach.
How Foundayo Works in Your Body
-
The Ingestion: You swallow the pill. (With a morning coffee, after a big lunch, or right before bed—it doesn’t matter).
-
The Absorption: The small molecule bypasses stomach degradation and enters the bloodstream.
-
The Activation: It binds tightly to the GLP-1 receptors in the pancreas and brain.
-
The Result: You feel profoundly full on smaller portions, cravings are blunted, and your metabolic engine is stabilized.
Read more article Expert: Why Early Intervention is Key on Autism Awareness Day
📊 Foundayo vs. The Competition: 2026 GLP-1 Landscape
How does the new kid on the block stack up against the titans of the industry? Let’s break it down.
| Feature | Foundayo™ (Orforglipron) | Wegovy® (Semaglutide) | Zepbound® (Tirzepatide) | Rybelsus® (Semaglutide) |
| Delivery Method | Daily Pill | Weekly Injection | Weekly Injection | Daily Pill |
| Active Ingredient | Non-peptide GLP-1 | GLP-1 | GLP-1 / GIP | GLP-1 |
| Food/Water Rules | None | N/A | N/A | Strict 30-min fasting |
| Average Weight Loss | ~14.7% (at 36 weeks) | ~15% (at 68 weeks) | ~20.9% (at 72 weeks) | ~5-7% (standard dose) |
| Main Advantage | Ultimate convenience | Proven long-term data | Highest efficacy | First to market oral |
(Data based on 2025/2026 FDA trial summaries and clinical findings)
📈 The Phase 3 ACHIEVE Trials
Before the FDA stamped its approval, Foundayo had to prove it wasn’t just convenient, but highly effective. The pivotal Phase 3 trials (completed in late 2025) showcased remarkable numbers.
The Setup: A randomized, double-blind, placebo-controlled trial involving over 3,000 adults with obesity (BMI ≥30). The Execution: Patients took varying doses of Foundayo once daily for 36 weeks, without altering their baseline meal schedules. The Results:
-
Weight Reduction: Patients on the highest dose (45mg) saw an average weight reduction of 14.7%.
-
Cardiometabolic Health: Significant improvements in blood pressure, fasting lipids, and waist circumference.
-
Tolerability: The side effect profile matched injectable GLP-1s (mild to moderate nausea, mostly dissipating after the titration period).
Consider “Sarah,” a 42-year-old traveling sales director who participated in the trials. Traveling through airports with refrigerated injectables was a nightmare for her. The strict fasting rules of other pills made her morning routine impossible. Foundayo allowed her to drop 32 pounds simply by adding a pill to her existing daily vitamin regimen.
⚖️ Pros and Cons of Foundayo
The Pros:
-
Unmatched Convenience: No needles, no refrigeration, no timers, no water limits.
-
High Efficacy: Competes directly with injectable Wegovy in total weight loss percentage.
-
Supply Chain Resilience: Small molecule pills are easier and cheaper to manufacture at scale than complex biologics (injectables), potentially easing the chronic shortages seen in 2023–2025.
-
Discreet: Taking a pill in public is entirely normalized.
The Cons:
-
Gastrointestinal Adjustment: Like all GLP-1s, the first few weeks can cause nausea, diarrhea, or constipation.
-
Daily Routine Required: You have to remember to take it every day, unlike a weekly shot.
-
Cost: While potentially cheaper to produce, introductory pricing remains in the premium tier (insurance coverage pending for many providers).
More update official webside FDA Approval Press Release and the New England Journal of Medicine (NEJM) study on orforglipron Phase 3 efficacy.
💼 The Market Impact: Eli Lilly’s 2026 Dominance
From an investor standpoint, Foundayo is a masterstroke. Eli Lilly already captured the high-efficacy market with Zepbound (tirzepatide). Now, with Foundayo, they are capturing the convenience market.
Industry analysts project that the oral GLP-1 market will expand the total addressable obesity market by up to 40%. Why? Because millions of patients who suffer from trypanophobia (fear of needles) or simply refuse the hassle of injections will now enter the treatment funnel.
Eli Lilly is positioning Foundayo not just as a standalone treatment, but as a maintenance therapy. Once a patient reaches their goal weight on Zepbound, they can transition to the Foundayo pill to maintain their results indefinitely.
Do I need to take Foundayo on an empty stomach?
No. Unlike older oral GLP-1 medications, Foundayo has no food, water, or fasting restrictions. You can take the pill at any time of day, with or without meals, making it the most convenient GLP-1 currently available.
How much weight can you lose on Foundayo?
Based on Phase 3 clinical trials, patients taking the highest therapeutic dose of Foundayo (45mg) achieved an average body weight reduction of approximately 14.7% over a 36-week period, rivaling the results of popular injectable treatments.
Is Foundayo an injection or a pill?
Foundayo is a once-daily, oral pill. It completely eliminates the need for subcutaneous injections and does not require refrigeration, making it ideal for patients who travel frequently or have an aversion to needles.
Does Foundayo replace Zepbound or Wegovy?
Not necessarily. Foundayo offers a highly effective oral alternative for those who dislike injections. However, dual-agonists like Zepbound (tirzepatide) may still offer slightly higher total weight loss percentages for patients requiring maximum metabolic intervention.
The FDA approval of Foundayo (orforglipron) isn’t just a minor update to the pharmaceutical playbook; it is a seismic shift in how we treat obesity. By stripping away the needles, the fasting, and the rigid scheduling, Eli Lilly has democratized access to top-tier metabolic care.
For the first time, achieving profound, medically-assisted weight loss is as simple as taking your morning multivitamin. As we move deeper into 2026, Foundayo is poised to not only dominate the market but fundamentally change millions of lives for the better.
What are your thoughts on the new Foundayo pill? Would you switch from an injection to a daily pill? Drop a comment below, and don’t forget to share this guide with anyone navigating their metabolic health journey!

